
What is a core group element in Period 6? These are polonium, oxygen, sulfur, selenium and tellurium. One might also ask how many valence electrons does group 6a have? 2 valence electronsWhat is the other name of the 6a answer com group? 
Oxygen is paramagnetic because there are unpaired electrons in the O2 molecules. One might also ask what are the common oxidation states of the elements of group 6a?įor elements of group 6A, the most common oxidation numbers are 4, 6 and 2. Depending on where you look, this group is also known as Group 16 or Group VIA, which all mean the same thing. Polonium is one of the five elements belonging to chalcogens, or elements of group 6A, which include oxygen (O), sulfur (S), selenium (Se), tellurium (Te) and polonium (Po). How are the elements of group 6a similar? 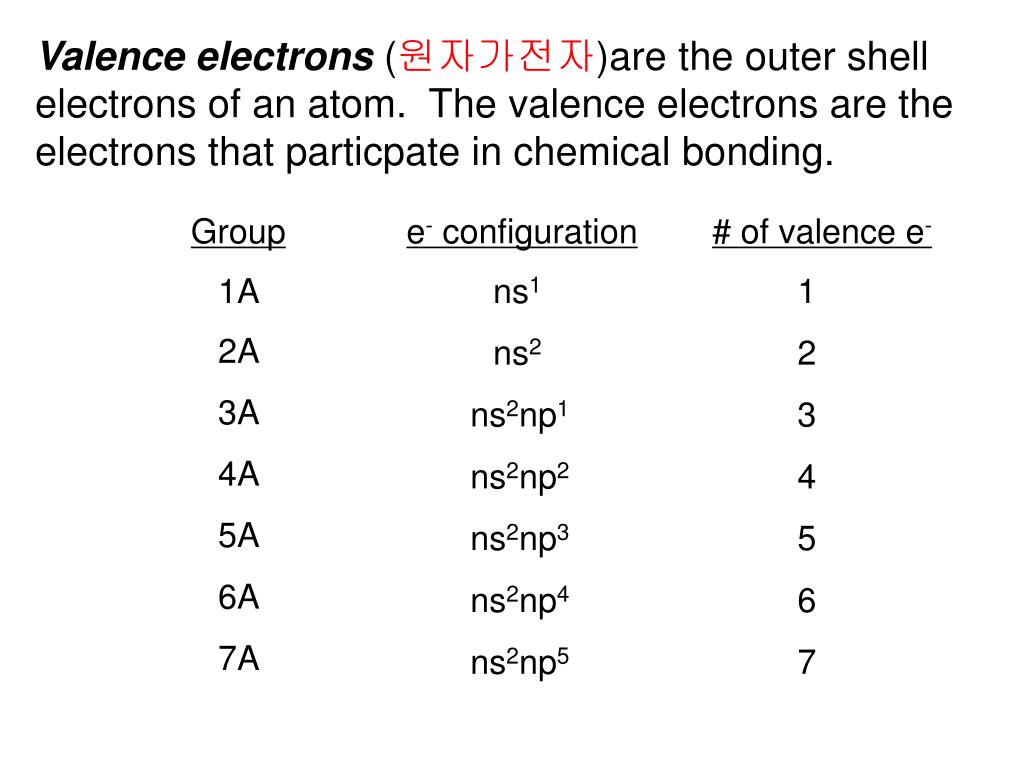
Group 6A (or VIA) of the periodic table includes chalcogens: non-metallic oxygen (O), sulfur (S) and selenium (Se), nonmetal tellurium (Te) and metallic polonium (Po). Group 6a Periodic Table Is it a Group 6a non-metal?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
